Lattice energy of nacl3/12/2023 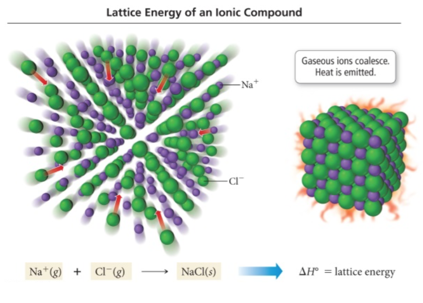
In equilibrium without energy supply, directional motion cannot be sustained without violating the laws of thermodynamics. Out of 200 people sampled, 94 had children.To impart directionality to the motions of a molecular mechanism, one must overcome the random thermal forces that are ubiquitous on such small scales and in liquid solution at ambient temperature.1) A car initially moves at 60 mph and then slows down to 45 mph.A coin is tossed three times and the events are shown as.Your roommate Pat decides to help with the party. When the following oxidation-reduction reaction is balanced inĪcidic solution.Six people A, B, C, D, E, and F, have decided to go to the.An automobile manufacturer claims that a new Hybrid model will.What is the pH of a solution prepared by mixing 25.0 mL of 0.100.ICO Given the following enthalpies, cal Enthalpies (kJ/mol): howing enthalpies, calculate the lattice energy of Nal and CsCls), respectively. ICO Given the following enthalpies, cal Enthalpies (kJ/mol): howing enthalpies, calculate the. 2) From the following data in the Born-Haber cycle, Na(s) → Na(g) 4C12(g) → Cl(g) AH:-108.Ģ) From the following data in the Born-Haber cycle, Na(s) → Na(g) 4C12(g) → Cl(g) AH:-108 kJ/mol AHj - 495.9 kJ/mol ara =-349 kJ/mol Na(g) → Na+(g) + e- Cl(g) + e-→ Cl-(g) Na(s) +4C12(g) → NaCl(s) AHoverall =-411 kJ/mol calculate the lattice energy of NaCI.Ģ0: Make use of the following information.Ģ0: Make use of the following information for calculating the electron Affinity of C AH = -216 kJ A H2 249 kJ AH3 = + 240 kJ A H4 ? A H5 786 kJ A Ho-411 kJ 1: 2Na 2: ½ Na(g 3: Cl2() 4: Cl eg) 5: Na () Cl 6: Nas 2Nas ½ Na () 2Ck Deposition Energy Ionisation Energy Atomization Energy Electron affinity Lattice Energy Standard Enthalpy of.Theīond energy of Cl2 is BE = 243 kJ/mol. The electron affinity of Cl is ΔH EA = -349 kJ/mol. The first ionization energy of Li is IE 1 =ĥ20 kJ/mol. The lattice energy of LiClĬonsider the following information. Use the following data to calculate the lattice energy (U) of NaCl(s) from sodium me chlorine: Enthalpy of formation (4H) for NaCl(s) -411 kJ/mol Enthalpy of sublimation (4Hub) of Na 107.3 kJ/mol The first ionization energy of Na (E,)-495.8 kJ/mol The bond dissociation energy (D) of Clh- 243 kJ/mol The electron affinity of Cl (Eea)- 348.6 kJ/mol. Use the following data to calculate the lattice energy (U) of NaCl(s) from sodium me.Ģ. Calculate the heat produced when 700.0 mL of 0.500 M HCl is mixed. Consider the reaction: 2HCl) + Ba(OH)2(aq) → BaCl(aq) + 2 H200 AH-118 kJ/mol a. Calculate AH (expressed in kJ/mol) for the following reaction based on the thermochemical information given: Na +Crce → NaCl AH ? Nas) + 2Cl2(g) → Nap + Cle AH = 230 kJ/mol Nag) + Cl → Na(+ Cl® AH = 147 kJ/mol Na(s) + Cl2(e) → NaCl) AH = -411 kJ/mol 3. Calculate AH (expressed in kJ/mol) for the following reaction based on the thermochemical information given.Ģ. QUESTION 13 Based off the following data, what is the lattice energy of sodium chloride? AHsub of Na = 108 kJ/mol Do of Cl2= 243 kJ/mol IE of Na(g) = 496 kJ/mol EA of Cl(g) = -349 kJ/mol AHf of NaCl = -411 kJ/mol O A. QUESTION 13 Based off the following data, what is the lattice energy of sodium chloride? AHsub.Determine the enthalpy of formation, AHf, for NaCl(s). The bond energy of Cl, is BE = 243 kJ/mol.The electron affinity of Cl is AHEA = -349 kJ/mol.The first ionization energy of Na is IE1 = 496 kJ/mol.The enthalpy of sublimation of Na is AHsub = 107.5 kJ/mol.The lattice energy of NaCl is AHlattice = –788 kJ/mol. 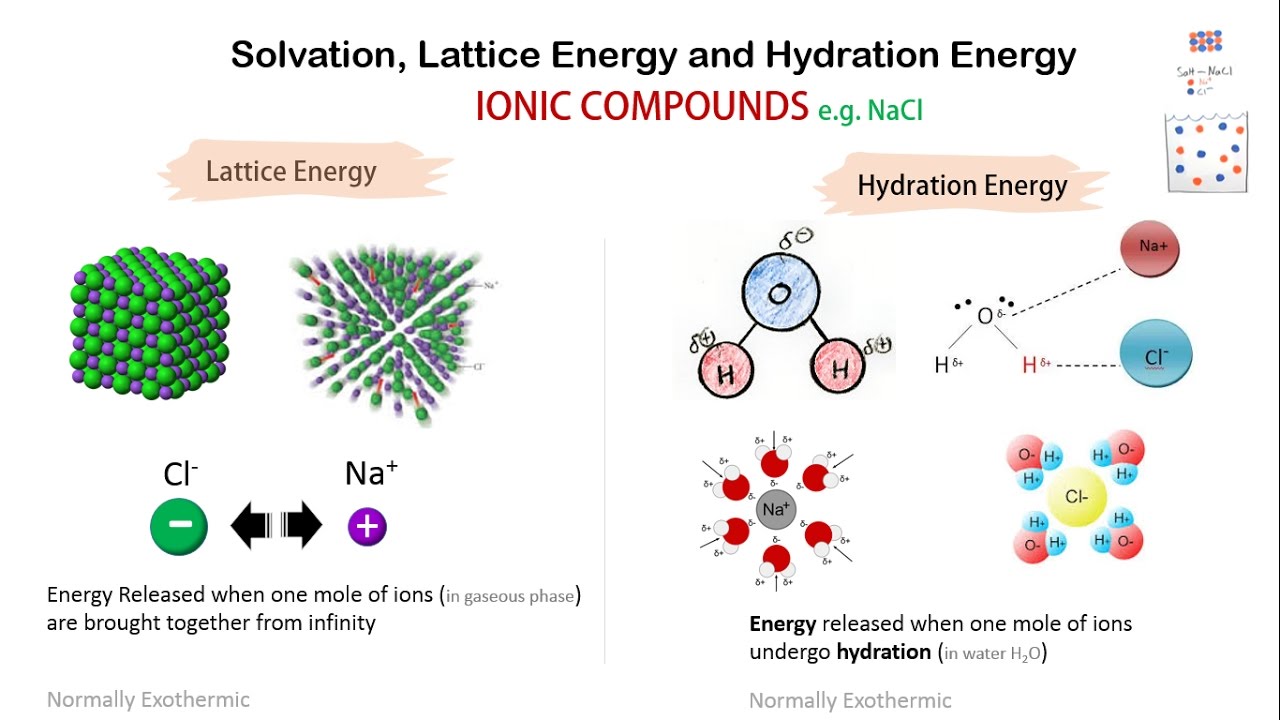
The electron affinity of Cl is ΔHEA=−349 kJ/mol.ĭetermine the enthalpy of formation, ΔHf, for NaCl(s). The first ionization energy of Na is IE1=496 kJ/mol. The enthalpy of sublimation of Na is ΔHsub=107.5 kJ/mol The lattice energy of NaCl is ΔH lattice=−788 kJ/mol
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
